Research
(i) Generation of cell type diversity in the vertebrate retina
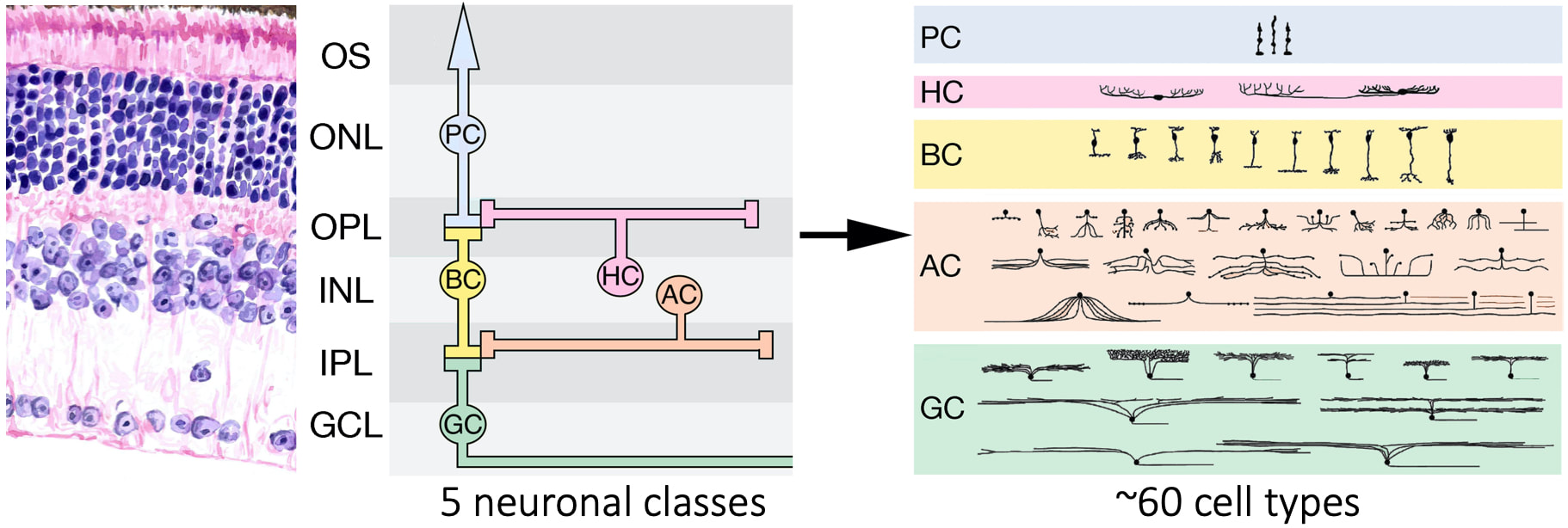
The retina is a thin, sensory neuronal tissue that lines the inside of the eye. Light-detecting neurons in the retina called photoreceptors (PC) convert the energy of light into a molecular signal. Before being sent to the brain, these signals pass through a region of the retina called the inner retina. Here, cells called interneurons (BC, AC, GC) form parallel circuits that extract and process different types of visual information. Although the retina has a relatively simple organization, it has a tremendous amount of cellular diversity that underlies its ability to parse out visual information. We are interested in understanding the cell intrinsic and extrinsic mechanisms that specify retinal cell type diversity. We use a variety of genetic, cellular and molecular approaches to understand these processes.
(i) Generation of cell type diversity in the vertebrate retina
The retina is a thin, sensory neuronal tissue that lines the inside of the eye. Light-detecting neurons in the retina called photoreceptors (PC) convert the energy of light into a molecular signal. Before being sent to the brain, these signals pass through a region of the retina called the inner retina. Here, cells called interneurons (BC, AC, GC) form parallel circuits that extract and process different types of visual information. Although the retina has a relatively simple organization, it has a tremendous amount of cellular diversity that underlies its ability to parse out visual information. We are interested in understanding the cell intrinsic and extrinsic mechanisms that specify retinal cell type diversity. We use a variety of genetic, cellular and molecular approaches to understand these processes.
(ii) Stargardt disease
Stargardt disease (STGD1) is an autosomal recessive disease with primarily juvenile onset that affects approximately 1:10,000 people and results from mutations in the ABCA4 gene. Although supportive care including the use of sunglasses and reduced vitamin A diets can help slow progression, there are no approved therapies, and all patients are expected to reach legal blindness.
Stargardt Disease pathology is strongly associated with the activation of a branch of our innate immune system called the complement system. The current model is that ABCA4 mutations lead to the accumulation of toxic products of the visual cycle called bisretinoids in the retinal pigmented epithelium. This is thought to cause localized oxidative stress that triggers activation of the complement system. This, in turn, leads to a pro-inflammatory response that results in progressive cell death and visual loss.
The requirement of complement system activation for Stargardt disease pathology has not been definitively demonstrated. We are addressing this by performing a "rescue” experiment to determine whether the loss of complement factor D (CFD), a gene which encodes a key rate-limiting protein involved in complement system activation, can block disease progression in a genetic Abca4 Stargardt model. This study will provide important proof-of-principle information necessary for the design of therapeutic approaches that we plan to pursue.
Stargardt disease (STGD1) is an autosomal recessive disease with primarily juvenile onset that affects approximately 1:10,000 people and results from mutations in the ABCA4 gene. Although supportive care including the use of sunglasses and reduced vitamin A diets can help slow progression, there are no approved therapies, and all patients are expected to reach legal blindness.
Stargardt Disease pathology is strongly associated with the activation of a branch of our innate immune system called the complement system. The current model is that ABCA4 mutations lead to the accumulation of toxic products of the visual cycle called bisretinoids in the retinal pigmented epithelium. This is thought to cause localized oxidative stress that triggers activation of the complement system. This, in turn, leads to a pro-inflammatory response that results in progressive cell death and visual loss.
The requirement of complement system activation for Stargardt disease pathology has not been definitively demonstrated. We are addressing this by performing a "rescue” experiment to determine whether the loss of complement factor D (CFD), a gene which encodes a key rate-limiting protein involved in complement system activation, can block disease progression in a genetic Abca4 Stargardt model. This study will provide important proof-of-principle information necessary for the design of therapeutic approaches that we plan to pursue.